Research News
A summary of the main, ongoing areas of research into treatments for PWS can be found on our Potential Future Treatments page.
LATEST DEVELOPMENTS
Vagus nerve stimulation for behaviour in PWS
SEPT 2022 - A focused campaign by the Foundation for Prader-Willi Research (#VNS4PWS) has been raising funds toward a large-scale trial of VNS which will be conducted across multiple centres in the USA. This trial requires quite significant funding, but it is hoped that this use of research funds could be the most impactful to date. So, what is vagus nerve stimulation? VNS alters…
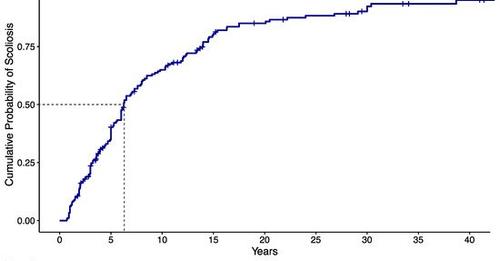
High prevalence of scoliosis in large cohort of patients
MARCH 2022 - The prevalence rate of scoliosis in PWS has not been well established with figures ranging between 40% to 80% of patients in various studies and surveys. It is known that scoliosis typically increases with age, with occurrence data ranging from 23% of children with PWS before 4 years, to around 70% of adults with PWS. The purpose of this Italian study…
A new trial is required for carbetocin / LV-101
JAN 2022 - A New Drug Application for LV-101 (intranasal carbetocin) was granted FDA Priority Review last year for the treatment of hyperphagia and behavioural symptoms in PWS, but ultimately, the review was unsuccessful as there were concerns about efficacy. Following the positive results observed on hyperphagia, anxiety and distress symptoms at the lower 3.2mg dose in the phase 3 trial (CARE-PWS) and during…
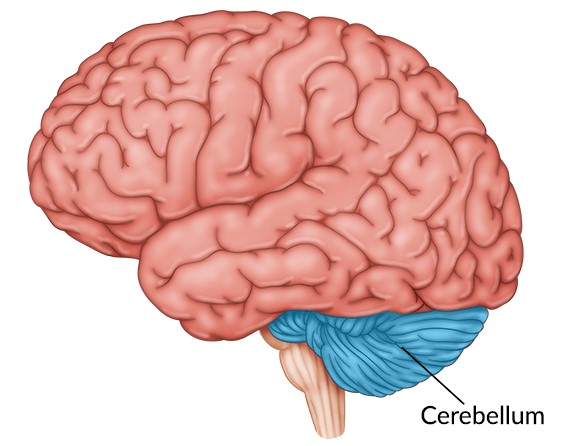
New area of the brain identified for developing therapeutic targets to manage hyperphagia
NOV 2021 - The hypothalamus and hindbrain have previously been implicated in appetite regulation, but drugs targeting these areas have not been as successful as hoped. Dr. J. Nicholas Betley, of the University of Pennsylvania, and Dr. Albert Chen, of the Scintillon Institute, speculated that the cerebellum could play a role in hunger suppression and reached out to Laura Holsen, who had a rare…
DCCR development update (DESTINY PWS and extension studies)
SEPT 2021 - DCCR (Diazoxide Choline Controlled Release) is a once-daily treatment for hyperphagia and body composition being developed by Soleno Therapeutics for the PWS community. DCCR is thought to affect leptin pathways by opening KATP channels, and also to improve GABA signaling. (Leptin is a hormone that helps to regulate energy balance by inhibiting hunger and its pathway is thought to be disrupted…
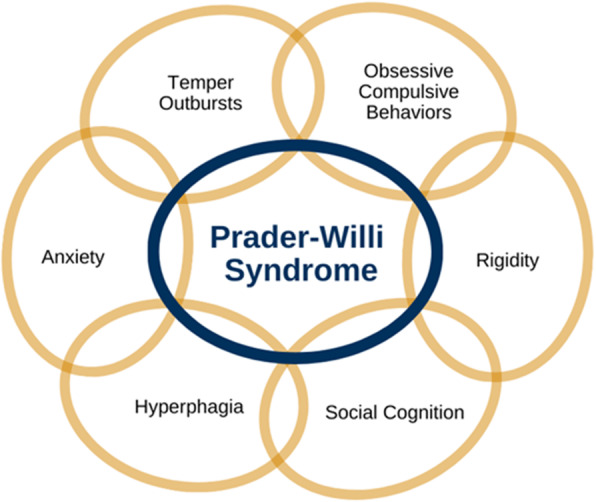
Consensus paper on behavioural features of PWS published by expert consortium
JUNE 2021 - The International PWS Clinical Trials Consortium have produced consensus definitions and descriptions of the key behavioural features of PWS, with the aim of providing standardised measures for treatment studies. FPWR have produced the infographic to the right which summarises the 6 definitions (a useful resource to print.) This consensus paper was produced because the lack of consensus in the field regarding…
Pharmacodynamic gene testing should be considered before initiating pharmacotherapy in PWS
NOV 2020 - It is known that side effects of psychotropic medications are often atypical in PWS compared to the general population, which is why a lower dose is often recommended to avoid the potential of adverse effects. This may be because medications are metabolised differently, or due to neurosensitivities. Psychtropic treatments can sometimes be counter effective and can result in the worsening of…
Top-line phase 3 results announced for Carbetocin trial – positive results at low dose
AUG 2020 - Oxytocin (OT) is a naturally occurring hormone released from the hypothalamus in the brain. It is associated with maternal care, bonding, feeding in infancy, social cognition, OCD, anxiety and appetite control. Carbetocin is an oxytocin-like drug which is thought to positively affect appetite and emotion because of the decreased levels of oxytocin hormone in PWS, thought to contributing to the behavioural alterations…
Accelerated ageing in PWS
NOV 2019 - With better management, people with PWS are living longer today, but the number of people known to our Association aged over 50 years is still relatively small. There is hope that this will change in the future with the advantages of early diagnosis and growth hormone treatment, but it has long been hypothesised that premature ageing occurs in PWS. It has…
GABA – a new potential therapeutic target for challenging behaviour
AUG 2019 - Can a GABA-modulating drug normalise the balance of neurotransmitters in the brain and improve behaviour in PWS? GABA (gamma aminobutyric acid) is an amino acid acting as a brain neurotransmitter (chemical messenger.) GABA inhibits certain brain signals and decreases activity in the nervous system which results in a calming effect. When GABA attaches to a brain protein known as a GABA…
Pitolisant data published and film of patient experiences launched
MAY 2019 - The case series data from 3 children trialling pitolisant has been published. It was noted that the treatment was well tolerated and in addition to the previously reported positive effects on cognition (mental clarity and processing speed), pitolisant had the additional benefit of normalising the children’s relationship to food. You can read more details about the dosing and any side effects in…
Characteristics of temper outbursts in Prader-Willi syndrome
NOV 2018 - In this study, Drs. Rice, Einfeld and Woodcock aimed to develop a comprehensive understanding of temper outbursts in PWS. A survey was developed from interviews conducted with individuals with PWS and their caregivers. The survey was completed by 101 primary caregivers. The findings suggest that outburst frequency decreases with age while duration increases. Adolescents exhibited more severe behaviours than children or adults.…
Cessation of GH therapy and rapid increase in visceral fat (deep abdominal fat located around internal organs) and LDL cholesterol
SEPT 2018 - A Japanese research team, Koizumi et al, used dual-energy X-ray absorptiometry (DEXA scans) and abdominal computed tomography (CT scans) to compare the fat distribution between before ceasing GH treatment and 6 months and 12 months after the cessation of GH therapy in 7 adult PWS patients. All patients had been treated with GH for an average of 14.7 years and discontinued…
Phase 2 carbetocin results published, confirming positive results for hyperphagia and behaviour
JUNE 2018 - Results of the phase 2 carbetocin trial (oxytocin-like nasal spray) have been published and phase 3 is expected to begin later this year. Carbetocin is similar to oxytocin, but specifically targets only the oxytocin receptor which may reduce impact on other systems, i.e. vasopressin signalling, and is therefore thought to limit any potential unwanted side effects. (Activation of vasopressin receptors could…
Hypothalamus analysis characterized by neuronal loss, altered neuro-plasticity and neuroinflammation – a potential role for BDNF identified
MARCH 2018 - For cell building and maintenance, DNA ‘instructions’ are ‘read’ and transcribed into RNA and these ‘read-outs’ are called transcripts. A transcriptome is the complete set of RNA transcripts that are produced by the genome (all the genetic material in a cell). In this study, transcriptomic analysis of hypothalamic PWS brain tissue by RNA sequencing was performed. Altogether, 3676 differently expressed genes were…
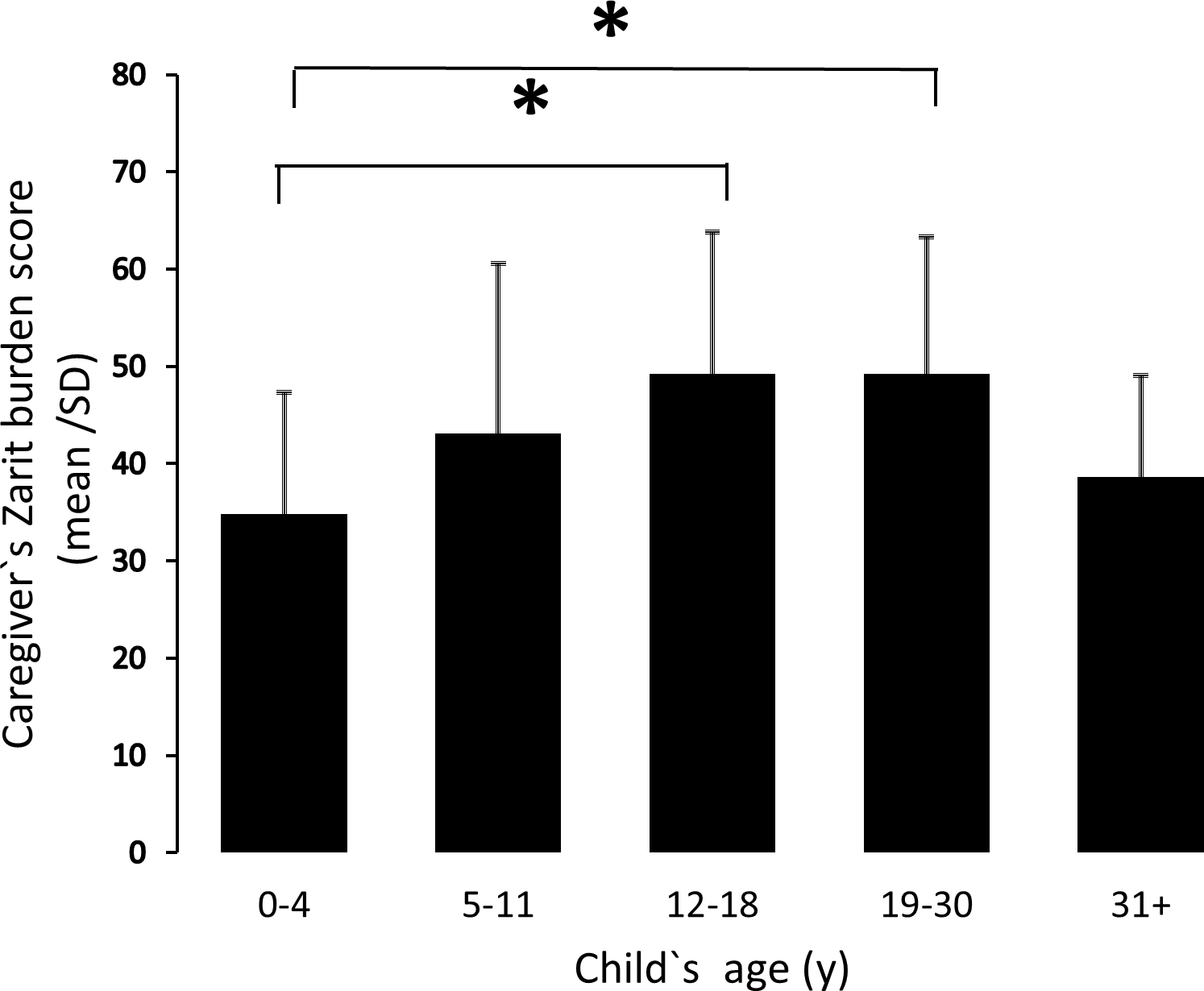
High levels of caregiver burden measured in Prader-Willi syndrome
MARCH 2018 - This study aimed to measure levels of caregiver burden and explore the impact of PWS on caregiver quality of life by examining burden in 142 caregivers (predominantly mothers) of children and adults with PWS living in the USA using the Zarit Burden Interview. Results showed that caregivers experienced strikingly high caregiver burden with ZBI scores being highest for caregivers of teenage and…
DCCR benefits for PWS confirmed in Magel2 deficient mice with improved endurance capacity
FEB 2018 - Diazoxide Choline Controlled Release (DCCR) is currently being developed by Soleno Therapeutics following a successful phase 2 trial in PWS patients which observed improvements in hyperphagia, a reduction of body fat and increase in lean body mass. They are advancing DCCR into a phase 3 clinical development program which is due to start recruiting from March 2018. A newly published study at the University of Alberta tested whether chronic…
Aberrant, autistic, and food-related behaviours in PWS – comparisons between adolescents, young adults and adults.
FEB 2018 - These studies explored the differences of age as well as genotype in regards to the severity of behavioural symptoms in PWS, with emphasis on the comparison between the adolescent, young adult and adult age groups. Statistically significant differences were found in behaviour scores, with higher scores in young adults than adults (results) and higher scores in young adults than adolescents (results). Differences…
Australian Magel 2 research closer to waking dormant maternal PWS genes
JAN 2018 - News from the Prader-Willi Research Foundation of Australia: Associate Professor Marnie Blewitt at the Walter and Eliza Hall Institute of Medical Research (WEHI) has identified 3 well known medical compounds which are non-toxic, and all inhibit the activity of a protein known as SMCHD1. SMCHD1 normally keeps the maternal copy of MAGEL2 (a gene associated with PWS) and the neighbouring genes…
Snord116 research news from the Garvan Institute of Medical Research, NSW, Australia
OCT 2017 - Hypothalamus Specific Re‐Introduction of SNORD116 Increased Energy Expenditure The Snord116 gene cluster has been recognised as a critical contributor to PWS, with mice lacking Snord116 displaying many classical PWS characteristics, including low postnatal body weight, reduced bone mass and increased food intake. However, these mice do not develop obesity due to increased energy expenditure. To understand the physiological function of Snord116 better and…
CLINICAL TRIALS & SURVEYS (NZ)
PATH for PWS (Paving the way for Advances in Treatments and Health)
A study to help better understand serious medical events in PWS over a 4-year period, as well as evaluate how PWS related behaviours change over time. The data from this study is intended to inform the development and clinical trial design of potential new treatments. This is an online study with no visits required. The enrolled participant with PWS must be 5yrs+ and live in the USA, Canada, Australia or New Zealand. For the time and effort of participating in this study, participants will receive e-gift card vouchers for Amazon Australia. For more information and to enroll, visit www.pathforpws.com. Read More >
Mindfulness-Based Intervention for Temper Outbursts
Evaluating mindfulness-based intervention for managing temper outbursts and the impact on anxiety. Researchers at The University of Sydney are recruiting individuals with and without PWS, 13 to 30 years, who have a mild intellectual disability or typical IQ. Conducted remotely via phone, email and video-conference, the study is open to families within and outside of Australia. Primary caregivers will be trained to use and teach the intervention. A teacher or support worker will help monitor behaviours. Read More >
Oxytocin Research (Australia)
Recruiting individuals with and without PWS ages 13 to 30 years. It involves a 2 hour visit at either the University of Sydney in Camperdown, NSW or the Royal Children’s Hospital in Parkville, Victoria. Read More >
Liraglutide for weight management
As part of a multi-centre international trial, Prof Paul Hofman at Starship Hospital has been recruiting patients 12-18 years with BMI>95th percentile.
Read More
Liraglutide has an extensive safety record in adults and similar, albeit less, data in adolescents and children. Funding exists for patients and their family to travel to Auckland for study visits during a 12 month trial. Click to read further information about this trial. (Liraglutide is a GLP Receptor Agonist. GLP-1 is a gut derived hormone which reduces food intake and causes weight loss due to slowed gastric emptying and through direct central effects.) Contact: p.hofman@auckland.ac.nz
Sleep in genetic neurodevelopmental disorders
Researchers at the University of Canterbury investigating the effectiveness of treatments for sleep disturbance wish to gather information related to parents’ perceptions about their child’s sleep problems.
Read More
Open to parents of children of any age, so parents can complete for their adult child too. Participation is by a short (10-15 minute) online survey – survey link.
Contact: Laurie McLay. Ph (03) 369 3522
More about clinical trials…
More information about trials taking place specifically for PWS can be found here, and you are also able to search the Australian New Zealand Clinical Trials Registry and ClinicalTrials.gov
GLOBAL PWS REGISTRY
The purpose of the PWS Registry is to enhance the understanding of the full spectrum of PWS characteristics. The registry will also accelerate the completion of studies and clinical trials, plus determine areas of needed research and treatments.
Register to provide details on topics such as as developmental milestones, scoliosis, medications, appetite, behaviour and other clinical symptoms. You do not need to complete the entire registry in one sitting!